An Introduction to Bioconjugation
Bioconjugation: What Is It?
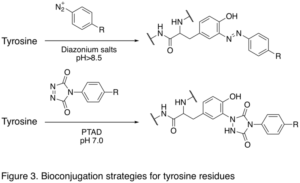 It is a chemical technique that’s used to couple two different molecules together with one being a biomolecule like a protein, carbohydrate, or nucleic acid. Of these, proteins are more diverse biomolecules since they contain a …
It is a chemical technique that’s used to couple two different molecules together with one being a biomolecule like a protein, carbohydrate, or nucleic acid. Of these, proteins are more diverse biomolecules since they contain a …
Raisha Rana
0